|
Assuming that the entropy of an ionic solid depends upon the magnitude of the ionic charges, Latimer obtained entropies of negative ions by subtracting entropies of positive (metal) ions (Me +n) from the experimental entropies of corresponding salts (e.g., Me +Cl, Me 2+Cl 2, Me 3+Cl 3). Latimer developed and additive method for the estimation of standard (absolute) entropies of solid compounds where entropies of solids are calculated as a sum of valence-dependent ionic contributions to the solid. Estimation methods for absolute entropies: S o 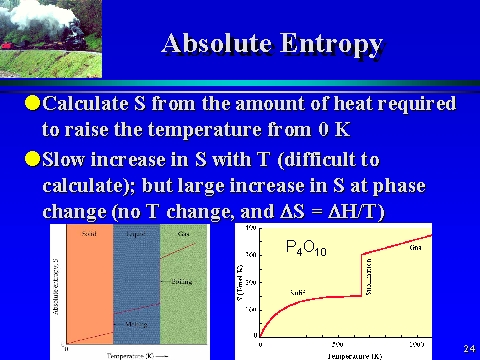
Such techniques are well established in the scientific literature to predict Gibbs energies and heats of formation, absolute entropies and heat capacities of solids. 
Therefore, various empirical and semi-empirical estimation techniques such as group contribution methods and family analyses were proposed in the literature to predict the properties of solid compounds. Absolute entropy is then calculated using the following equation:Ī precise and accurate thermodynamic measurement is always preferred over an estimation, but such measurements are not always possible because of many reasons, which include the solids that don’t exist at low temperatures (because of their phase transitions), hydrated solids existing only at specific ranges of temperatures, solids unstable at experimental conditions, etc. Enthalpies of formation are often derived from calorimetric measurements and absolute entropies can be calculated from cryoscopic heat capacity measurements starting from a very low temperature (closed to 0 K) to 300 K. Thermochemical properties such as heats of formation and heat capacities for many solid compounds can be measured. Heat capacity, C p o : The amount of heat that must be added to one unit of mass of the substance to cause an increase of one unit in temperature.ΔG f o = ΔH f o – T ΔS f o, where ΔS f o = S o – ΣS o elements Absolute entropy represents thermodynamic data that provide the link between enthalpy and Gibbs energy of formation: The third law of thermodynamics states that the entropy of a perfect crystal at absolute zero is exactly equal to zero. Absolute entropy, S o : The entropy change of a substance taken from absolute zero to a given temperature.Enthalpy of formation, ΔH f o : Change in enthalpy of formation when one mole of a solid is formed from its elements.Gibbs energy (or free energy) of formation, ΔG f o: Change in Gibbs energy of formation when one mole of a solid is formed from its elements.We will be dealing with the following thermochemical properties: It is necessary to know thermochemical properties (i.e., standard-state properties) of anhydrous and hydrated solid compounds to model phase equilibria in all, natural and industrial, chemical processes. AugChemistry What thermochemical properties are of most interest and why?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
